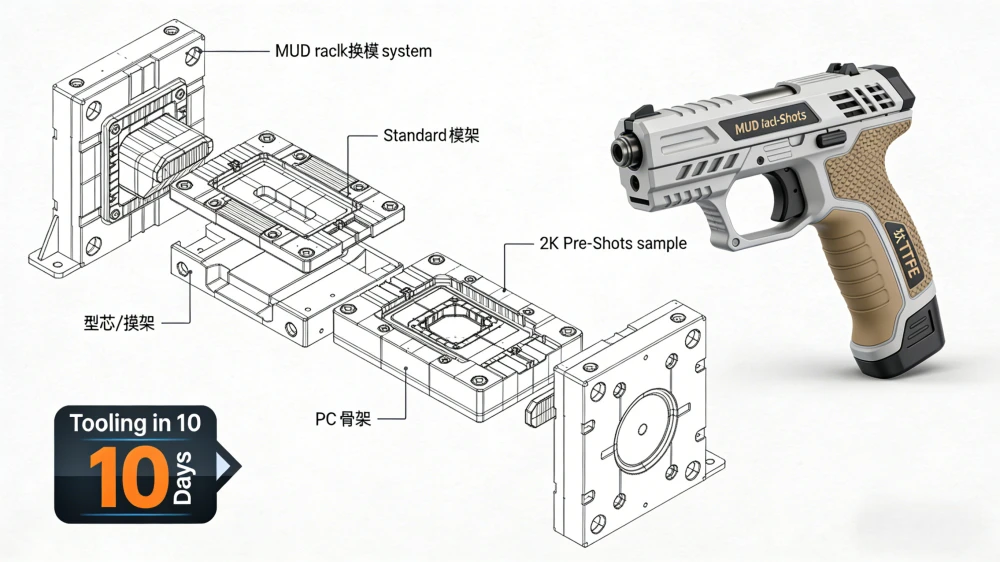
In the medical technology (MedTech) industry, there is zero margin for error. Whether it’s a diagnostic housing or a surgical tool component, the quality of your China injection molding factory directly impacts patient safety and regulatory approval.
Why ISO 13485:2016 Certification is Mandatory
At YG TeCH, we understand that medical device manufacturing requires more than just standard quality control. Our ISO 13485:2016 certification ensures:
Full Traceability: Every batch of raw material and every production run is documented.
Risk Management: Strict adherence to safety and efficacy standards throughout the manufacturing process.
Clean Production: Controlled environments to prevent contamination of sensitive medical parts.
Engineering Excellence for MedTech
Medical products often require advanced engineering materials and complex geometries. YG TeCH provides:
Tight Tolerances: We consistently achieve a precision of +/- 0.01mm for critical medical components.
LSR (Liquid Silicone Rubber): Ideal for medical-grade seals, valves, and wearable devices due to its biocompatibility and high stability.
High-Performance Polymers: Expertise in processing PEEK, PPS, and TPU for implantable-grade or chemically resistant applications.
Your Strategic MedTech Partner
From Low-Volume Trial Runs (NPI) to mass production, we offer a one-stop solution for product realization. Our expertise in IMD/IML and IM-3D-E also allows for the integration of electronics into sleek, sterilized medical housings.